Explain how hydrogen bonds form in water. How do these bonds influence properties of water? How
might the phase of water (liquid, solid, or gas) change the interaction of hydrogen bonds? What will be an ideal response?
ANS:Answer
should include:
? Hydrogen bonds form in water when the hydrogen atom of one water molecule is
attracted to the oxygen atom of another molecule. This is due to water being a polar
molecule which creates partially positive and negative portions of each molecule.
? Hydrogen bonds allow water molecules to link together. This allows water molecules to
stick to one another, a property called cohesion. Cohesion leads to high surface tension
for water. Hydrogen bonds also influence waters adhesion to solids as well. Both
cohesion and adhesion cause capillary actions to occur. These properties contribute to
water remaining a liquid at normal temperatures and pressures.
? Hydrogen bonds also are responsible for the color of water. The vibrations of these
bonds absorb red light energy while reflecting and scattering blue light.
? The phase of water has a large influence on the hydrogen bonds. When water is a solid,
the hydrogen bonds become compressed and very rigid as the water molecules begin to
stack. They eventually form in hexagonal patterns. The hydrogen bonds are unable to
hold when water transitions from liquid to a gas.
You might also like to view...
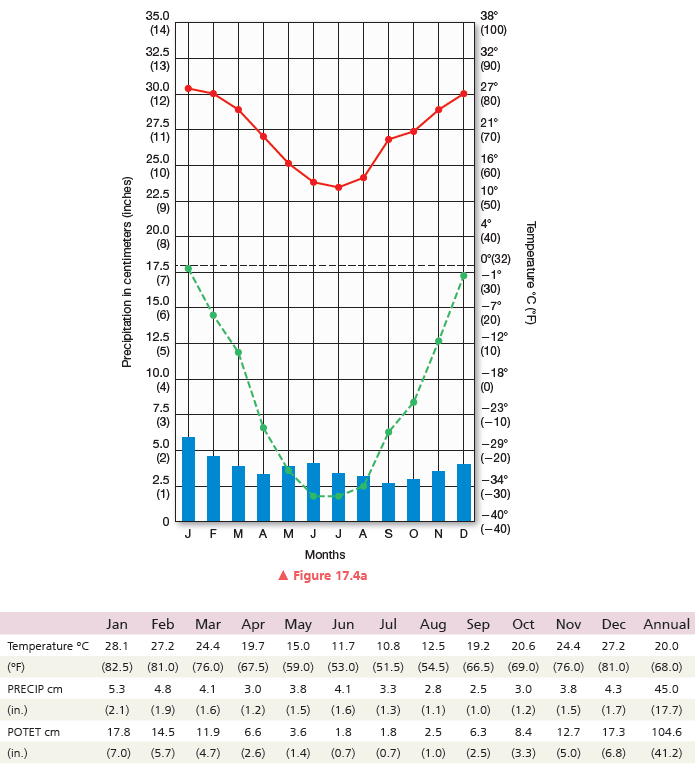
Annual temperature range:
The greatest planetary pool of nitrogen is ________
A) the lithosphere B) the hydrosphere C) in fossil fuel deposits D) the atmosphere E) the biosphere
The wet season of the tropical monsoon climate of SE Asia occurs during the season of ________ Sun due to the presence of the ________ over the continent. (low Sun = "winter" and high Sun = "summer")
A) low; ITCZ B) low; subtropical high C) low; Hadley cells D) high; subtropical high E) high; ITCZ
If you had this type of deformation of a region, it would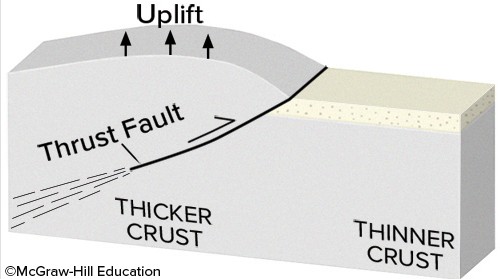
A. thin the crust and cause subsidence. B. thicken the crust and cause subsidence. C. thin the crust and cause uplift. D. thicken the crust and cause uplift. E. None of these choices are correct.