Based on their relative positions in the periodic table, which might you expect to be a stronger oxidizing agent, chlorine or fluorine? Why?
A. Chlorine should behave as a stronger oxidizing agent because it has a smaller effective nuclear charge in its outermost shell.
B. Fluorine should behave as a stronger oxidizing agent because it has a smaller effective nuclear charge in its outermost shell.
C. Fluorine should behave as a stronger oxidizing agent because it has a greater effective nuclear charge in its outermost shell.
D. Chlorine should behave as a stronger oxidizing agent because it has a greater effective nuclear charge in its outermost shell.
Answer: C
You might also like to view...
List the following compounds in order of least oxidized to most oxidized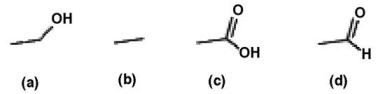
A. c < d < a < b B. a < b < c < d C. b < a < d < c D. b < a < c < d
Members of the mammal order Cetacea include desert-dwelling mammals that conserve water by
having thick skins. Indicate whether the statement is true or false.
In the history of the last 100 years, how is the automobile linked to urban sprawl?
What will be an ideal response?
Which of the following is true of the G-protein-adenylate cyclase cascade?
A) It is only associated with monoamine messengers. B) It is only associated with peptidergic messengers. C) It is associated with both monoamine and peptidergic messengers. D) It is associated with neither monoamine nor peptidergic messengers.