Why are the melting temperatures of most ionic compounds far greater than the melting temperatures of most covalent compounds?
A. Covalent bonds are not as strong as ionic bonds.
B. Most covalent compounds have at least one weak bond in their structure that is easily broken when heat is added.
C. Ionic bonds are so much stronger than the molecular attractions between covalently bonded compounds.
D. As a solid, salts have a very organized crystalline structure which takes a lot of energy to break apart.
Answer: C
You might also like to view...
When a developer buys land beyond the edge of growth causing the infrastructure network to be developed in an irregular pattern, it is called
A) suburban growth. B) urban sprawl. C) leapfrogging. D) agglomeration.
What is often found in the middle of manganese nodules?
a. Microtektites b. Shark teeth or bone fragments c. Living bacteria d. Sand grains
What will happen during movement on these normal faults?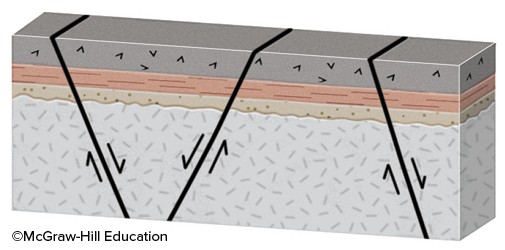
A. The fault blocks will rotate in the same direction, like books sliding on a shelf. B. The crust will thin and the region will be uplifted. C. The crust will thicken and the region will be uplifted. D. The blocks will be uplifted or downdropped with only minor rotation.
What is NOT included in a watershed?
A. overland flow B. streamflow C. valley bottom D. valley sides E. entire interfluves