In an aqueous solution that has not reached the saturation point, why won't the ions bond to create mineral crystals?
A) The opposite charges of the ions repel each other.
B) The ions are too close together to form a regular internal crystalline structure.
C) The motion of the dissolved ions keeps them from joining.
D) There are too few ions to make any minerals.
Answer: C
You might also like to view...
Which of the following does NOT characterized one of the three phases of the third agricultural revolution?
A) significant surplus production B) food manufacturing C) chemical farming D) mechanization
In August 2012, the average temperature at Location X was 90°F. Describe the climate in Location X.
A. Location X has a climate featuring wide seasonal variation. B. Location X has a warm climate. C. Location X has a hot climate. D. There is not enough information given to answer this question.
Which of the following is not true of minerals? They are(have):
a. crystalline. b. organic. c. naturally occurring. d. definite chemical composition. e. characteristic physical properties.
At what point on this water vapor saturation curve graph will water vapor be deposited on ice crystals while liquid water droplets will evaporate?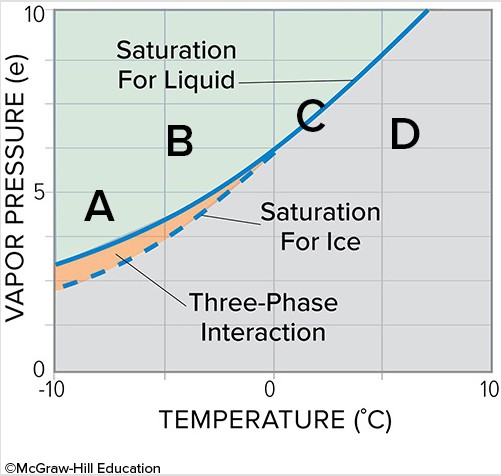
A. A B. B C. C D. D