Account for the observation that ethyl alcohol, C2H5OH, dissolves readily in water but dimethyl ether, CH3OCH3, which has the same number and kinds of atoms, does not.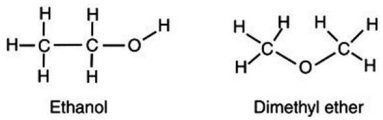
A. The high electronegativity of the carbon-oxygen-carbon bond on dimethyl ether creates a strong dipole charge on the ends of the molecule, making it highly soluble in water.
B. Because dimethyl ether lacks an -OH group, it is significantly less polar than is ethyl alcohol and is not readily soluble in water.
C. Because the carbons arrange themselves in a straight line, the ethanol can interact more easily with more water molecules, thus increasing its solubility.
D. The hydrogens on the dimethyl ether surround the molecule, shielding the inner atoms from interacting with the water.
Answer: B
You might also like to view...
Which of these regions is high because of mantle upwelling near a continental rift?
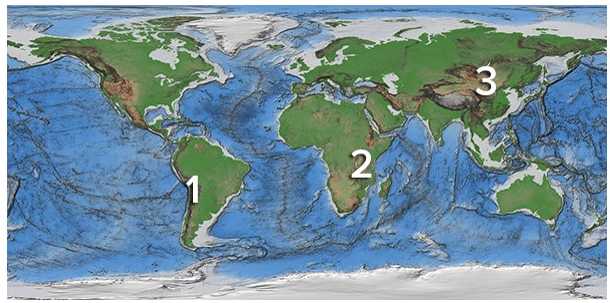
A) 1, Andes of South America
B) 2, East Africa
C) 3, Tibet
D) None of these are correct.
Geologists Vine, Matthews and Morley found striking similarities and symmetry in the pattern of _____ in rocks on opposite sides of ocean ridges.
A ) mineralization B ) density C ) gravity D ) magnetic residue
What is Papua New Guinea's most valuable export?
a. clothing b. workers c. minerals d. cassava e. sugar
Every day, meteorologists use their knowledge about how different air masses move in order to predict specific weather conditions for a given location. This is an example of ________ reasoning.
A. deductive B. inductive