Which of the following processes is the main cause of winter fog?
A. advection of cold air
B. uplift
C. adiabatic cooling
D. radiational cooling
Answer: D
You might also like to view...
Account for the observation that ethyl alcohol, C2H5OH, dissolves readily in water but dimethyl ether, CH3OCH3, which has the same number and kinds of atoms, does not.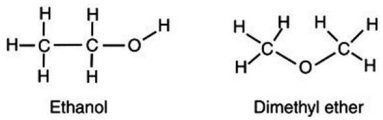
A. The high electronegativity of the carbon-oxygen-carbon bond on dimethyl ether creates a strong dipole charge on the ends of the molecule, making it highly soluble in water. B. Because dimethyl ether lacks an -OH group, it is significantly less polar than is ethyl alcohol and is not readily soluble in water. C. Because the carbons arrange themselves in a straight line, the ethanol can interact more easily with more water molecules, thus increasing its solubility. D. The hydrogens on the dimethyl ether surround the molecule, shielding the inner atoms from interacting with the water.
Which of the following is not a mineral?
A) volcanic glass B) ice C) quartz D) plagioclase feldspar
________ capture solar energy and use photosynthesis to produce sugars
A) Heterotrophs B) Secondary consumers C) Primary consumers D) Detritivores E) Producers
When considering both electron and H+ transport and the chemiosmotic pump, which of the following is the correct sequence of energy transformations?
A) chemical ? electrical ? mechanical B) chemical ? mechanical ? electrical C) electrical ? chemical ? mechanical D) electrical ? mechanical ? chemical