Why is the formation of iron hydroxide, Fe(OH)2, from Fe2+ and OH- not considered an oxidation-reduction reaction?
A. The iron is only oxidized and nothing is reduced.
B. The iron is only reduced, not oxidized.
C. Iron can never be oxidized or reduced.
D. The Fe2+ ion is not changed during this chemical reaction.
Answer: D
You might also like to view...
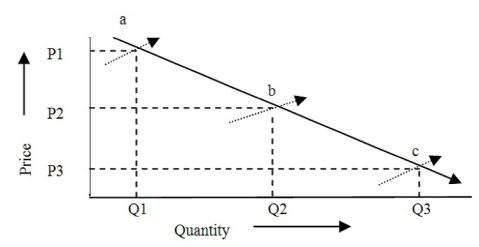 The graph shows how ________ when economies develop.
The graph shows how ________ when economies develop.
A. Quantities and prices naturally change through time B. Quantity increases as prices rise C. Prices fall as quantities increase D. Quantities and prices increase simultaneously E. Prices and quantities fall simultaneously
Explain why species found in cold habitats tend to be larger than related species found in hot habitats
What will be an ideal response?
The highest peak in the lower 48 states is located where?
A) The Sierra Nevadas B) The Rocky Mountains C) The Cascade Range D) The Tehachapi Mountains
The shoreline represents:
a. the general region of interaction between land and the ocean or lake. b. the average position of the water line. c. sea level. d. the average position of high tide. e. the exact, constantly changing contact between dry land and the standing water body.